Reconstitution Solution Water
$20.00
Reconstitution Solution Water
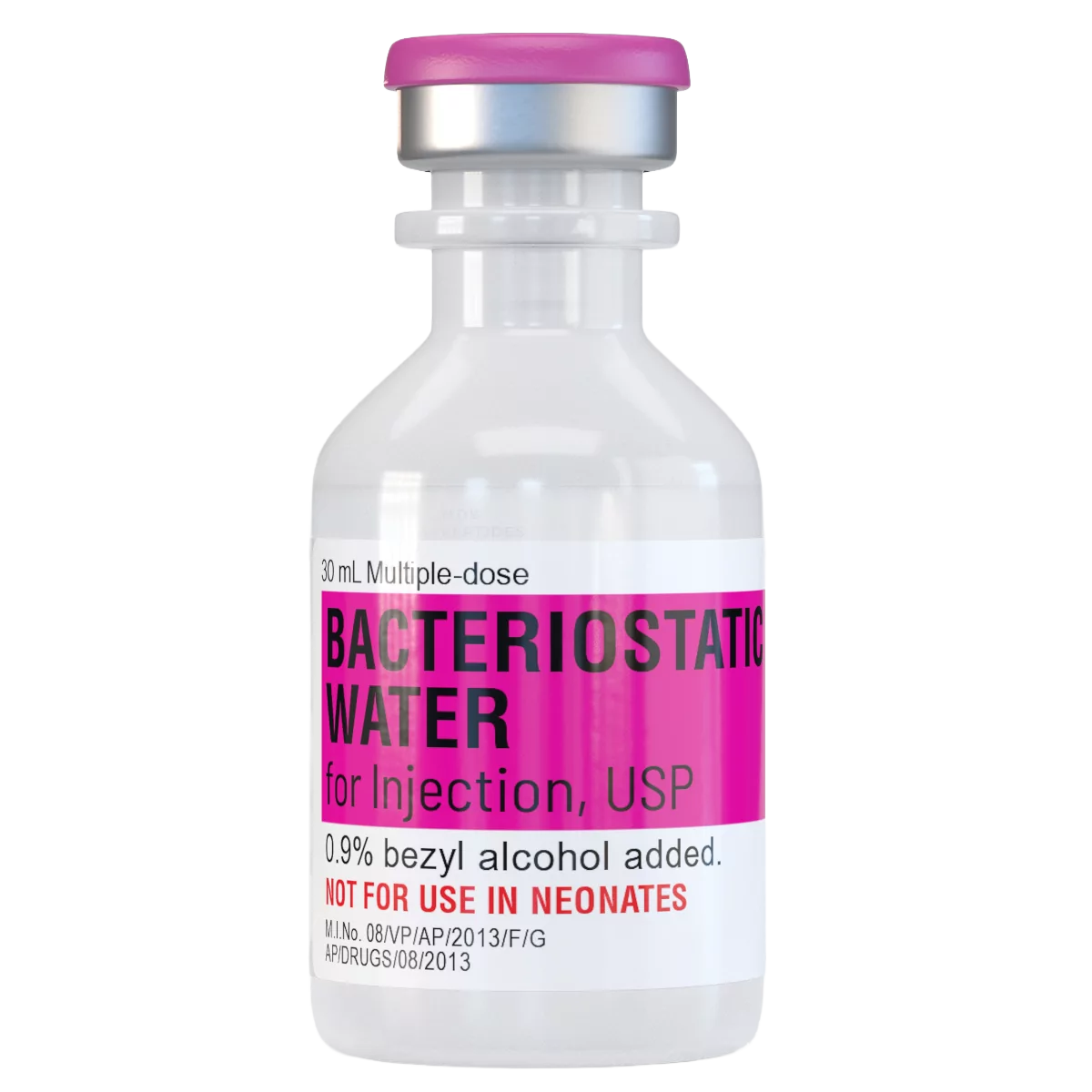
Bacteriostatic Water is a sterile, non-pyrogenic aqueous solution containing 0.9% benzyl alcohol as a bacteriostatic preservative. It is specifically formulated for use as a research diluent in laboratory and in-vitro settings where repeated withdrawals from a single vial are required. The presence of benzyl alcohol inhibits the growth of bacteria, allowing the solution to maintain sterility over multiple uses when handled under appropriate aseptic conditions.*
In research environments, bacteriostatic water is commonly used as a solvent or reconstitution medium for lyophilized compounds, including peptides, proteins, and other laboratory reagents intended strictly for experimental use.*
- Product Name: Bacteriostatic Water
- Composition: Sterile Water for Injection with 0.9% Benzyl Alcohol
- Volume: 30 mL multi-dose vial
- Form: Clear, colorless liquid
- Sterility: Sterile and non-pyrogenic
- Grade: USP
- Container: Sealed glass vial with rubber stopper
- Designation: For Research Use Only (RUO)
Scientific & Research Context
Bacteriostatic water is utilized in laboratory research as a controlled aqueous diluent where microbial inhibition is required without introducing reactive excipients. The inclusion of benzyl alcohol provides antimicrobial protection, making it suitable for studies involving repeated sampling, solution stability testing, and compound solubilization.*
In controlled research settings, bacteriostatic water is commonly used for:*
- Reconstitution of lyophilized peptides and proteins*
- Preparation of aqueous research solutions*
- Stability and degradation studies requiring preserved solvents*
- Laboratory workflows requiring multi-dose sterile access*
Its well-characterized composition and compatibility with a wide range of biomolecules make bacteriostatic water a standard reagent in pharmaceutical research, peptide chemistry, and molecular biology laboratories.*
Quality & Analytical Assurance
This bacteriostatic water is manufactured under stringent quality standards to ensure sterility, clarity, and consistency. Each vial is produced using USP-grade components and subjected to sterility and quality control testing to meet pharmaceutical-grade specifications for laboratory use.
Storage & Handling
Store bacteriostatic water at controlled room temperature, protected from excessive heat and light. Once accessed, proper aseptic technique should be used for all withdrawals to maintain sterility. Do not use if the solution appears cloudy, discolored, or if the vial seal is compromised.